64 SIS students get their COVID-19 vaccine shots
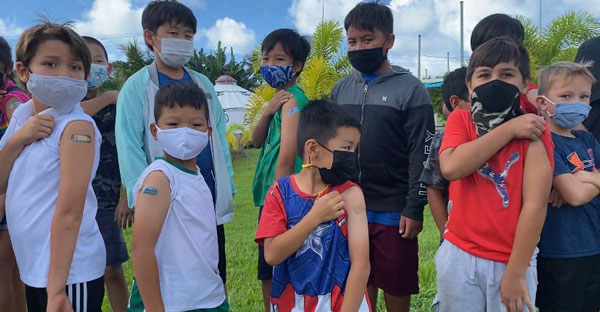
A total of 64 students aged 5-11 were vaccinated yesterday at Saipan International School as part of the Commonwealth Healthcare Corp.’s campaign to roll out its COVID-19 pediatric vaccinations at school sites.
CHCC is working with both the CNMI Public School System and some member schools of the Commonwealth Coalition of Private Schools as it kicks off its campaign to vaccinate students in the Commonwealth.
“We are extremely happy to be able to work with [CHCC] to offer the vaccination to families that want it. We decided to have a little fun with it by having our high school population welcome the vaccinated elementary students from a distance while playing music from the ‘Music to Get Vaccinated To’ playlist. I believe these types of proactive partnerships between [CHCC] and schools will help us maintain strong educational programs, while protecting our students in these difficult times,” said SIS headmaster Dr. Ron Snyder.
Other non-public schools have also signed up to be vaccination sites including, but not limited to, Brilliant Star Montessori School, Mount Carmel School, and Saipan Community School.
“Despite the unfortunate community spread over the past couple weeks, we as educators are grateful that the guidelines and protocols in place have mitigated the impact of COVID-19 at this time,” said Amanda Dunn, principal of Saipan Community School. “This also indicates how successful the COVID-19 Task Force, CHCC and the CNMI government have been in vaccinating our population and isolating individuals who have tested positive. I hope that the availability of the vaccine for children ages 5 – 11 will only strengthen our ability to get through this crisis together as a community.”
The CNMI received its first doses of the pediatric COVID-19 vaccine last Saturday and began rolling out immunizations that same day at the Pedro P. Tenorio Multi-Purpose Center.
The Pfizer vaccine has received approval from the U.S. Food and Drug Administration and the Centers for Disease Control and Prevention in the United States, joining many other countries worldwide offering the vaccine to younger children. For more information, visit: https://www.fda.gov/news-events/press-announcements/fda-authorizes-pfizer-biontech-covid-19-vaccine-emergency-use-children-5-through-11-years-age. (PR)






